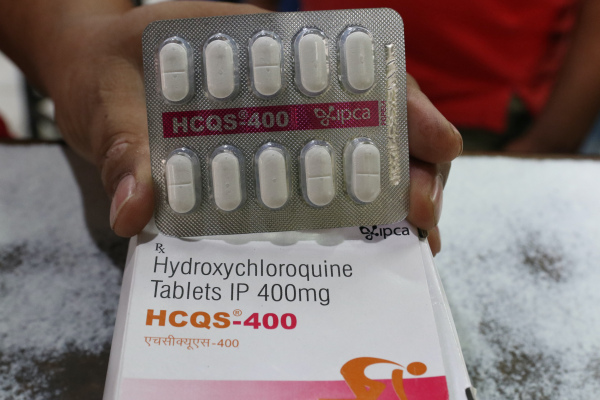
FDA revokes emergency authorization for chloroquine and hydroxychloroquine in COVID-19 treatment
TechCrunch·2020-06-16 05:55
The U.S. Food and Drug Administration (FDA) has revoked an emergency use authorization (EUA) that it previously issued for chloroquine and hydroxychloroquine, two anti-malarial drugs also used in the treatment of chronic rheumatoid arthritis (via The Washington Post). These are the drugs that President Trump famously touted as effective in COVID-19 treatment, despite major concerns raised with the scientific validity of early medical investigations that showed they were potentially effective against the infection beyond the ongoing global pandemic.
……Read full article on TechCrunch
America Health Technology International

One-stop lifestyle app dedicated to making life in Singapore a breeze!

Comments
Leave a comment in Nestia App